

We investigate pathomechanisms of diseases to improve diagnostics and treatment protocols based on mathematical modeling. Especially, we are working on quantitative descriptions of cellular pathways involved in cancer to support the development of improved therapeutic strategies in oncology. Inspired by the motto “semper apertus” of our university, we are always open for exchange and collaborations with other research groups across disciplines to move towards a functional understanding in pathophysiology and translational medical research.
The systems medicine team is dedicated to applications of mathematical modeling in medicine and cellular biology. Our aim is to understand functional aspects of signal transduction systems in cells with focus on cancer research. To this end, we combine different types of mathematical models with biological experiments or clinical data. To calibrate models, we apply experimental methods for protein quantification, microscopy as well as sequencing techniques. In particular, we perform live-cell imaging to quantify processes in single cells and capture heterogeneity in cell populations. Recently, we developed a microscopy platform for automated live-cell imaging and perfusion with chemotherapeutic drugs called CytoScanner. Using this platform and algorithms from the field of machine learning, we want to optimize administration schedules of chemotherapeutic drugs.
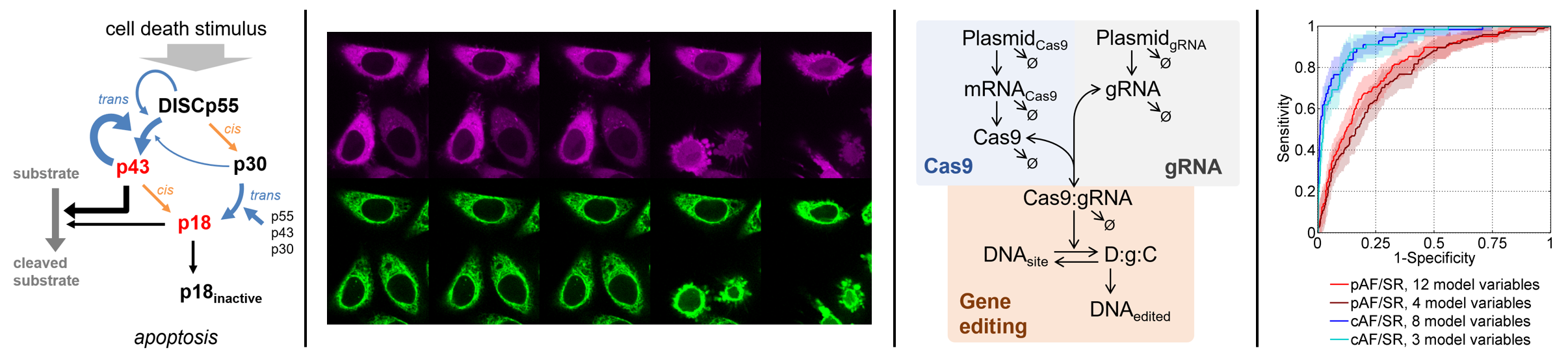
Furthermore, we are active on the field of data science in medicine. In this context, we work on developing predictive models for diagnostic applications and the early detection of diseases. Previosly, we established and clinically validated the ECHO-AF scores to predict the presence of paroxysmal atrial fibrillation (pAF) using non-invasive medical history and echocardiographic parameters

Dr. med. Dr. rer. nat. Stefan M. Kallenberger was educated as physician and physicist and works in the areas of systems medicine and systems biology. He is a member of the Hub for Innovations in Digital Health at Heidelberg University and the National Center for Tumor Diseases. His scientific work covers applications of mathematical models for understanding biochemical pathways, characterizing pathomechanisms, and for improving the early detection of diseases.
Dr. Dr. Stefan Kallenberger
Group leader at BioQuant-Zentrum
Adam L*, Stanifer M*, Springer F, Mathony J, Brune M, Di Ponzio C, Eils R, Boulant S, Niopek D#, Kallenberger SM#. Transcriptomics-inferred dynamics of SARS-CoV-2 interactions with host epithelial cells. Sci Signal. 2023;16: eabl8266. (*shared first authors, #corresponding authors)
Klein P*, Kallenberger SM*, Roth H, Roth K, Ly-Hartig TBN, Magg V, Aleš J, Talemi, SR, Qiang Y, Wolf S, Oleksiuk O, Kurilov R, Di Ventura B, Bartenschlager R, Eils R, Rohr K, Hamprecht FA, Höfer T, Fackler OT, Stoecklin G, Ruggieri A. Temporal control of the integrated stress response by a stochastic molecular switch. Science Advances 2022, 8, eabk202, (*shared first authors)
Schmidt C, Benda S, Kraft P, Wiedmann F, Pleger S, Büscher A, Thomas D, Wachter R, Schmid C, Eils R, Katus HA, Kallenberger SM. Prospective multicentric validation of a novel prediction model for paroxysmal atrial fibrillation. Clin Res Cardiol 2021
Aschenbrenner S*, Kallenberger SM*, Hoffmann MD, Huck A, Eils R, Niopek D. Coupling Cas9 to artificial inhibitory domains enhances CRISPR-Cas9 target specificity. Science Advances 2020;6:eaay0187, (*shared first authors)
Starkuviene V*, Kallenberger SM*, Beil N, Lisauskas T, Schumacher BS-S, Bulkescher R, Wajda P, Gunkel M, Beneke J, Erfle H. High-Density Cell Arrays for Genome-Scale Phenotypic Screening. SLAS Discov. 2019;24: 274–283, (*shared first authors)
Maier LJ*, Kallenberger SM*, Jechow K, Waschow M, Eils R, Conrad C. Unraveling mitotic protein networks by 3D multiplexed epitope drug screening. Molecular Systems Biology 2018;14:e8238, (*shared first authors)
Kallenberger SM, Unger AL, Legewie S, Lymperopoulos K, Klingmüller U#, Eils R#, Herten DP#. Correlated receptor transport processes buffer single-cell heterogeneity. PLoS Comput Biol. 2017;13: e1005779, (#corresponding authors)
Kallenberger SM, Schmid C, Wiedmann F, Mereles D, Katus HA, Thomas D, Schmidt C. A Simple, Non-Invasive Score to Predict Paroxysmal Atrial Fibrillation. PloS One. 2016;11: e0163621.
Kallenberger SM*, Schmidt C*, Dechent P, Forster C, von Steinbüchel N, Wüstenberg T, Strasburger H. Separating fusion from rivalry. PloS One. 2014;9: e103037. doi:10.1371/journal.pone.0103037, (*shared first authors)
Kallenberger SM, Beaudouin J, Claus J, Fischer C, Sorger PK, Legewie S#, Eils R#. Intra- and interdimeric caspase-8 self-cleavage controls strength and timing of CD95-induced apoptosis. Science Signaling 2014;7:ra23 (#joint senior authors, "Editors' Choice" in Science Vol 343, 2014),
The headline and subheader tells us what you're offering, and the form header closes the deal. Over here you can explain why your offer is so great it's worth filling out a form for.
Remember:
Feature Title
Lorem ipsum dolor sit amet
Consectetur adipiscing elit
Integer ornare lectus ut arcu
Tincidunt porttitor
Feature Title
Lorem ipsum dolor sit amet
Consectetur adipiscing elit
Integer ornare lectus ut arcu
Tincidunt porttitor
Feature Title
Lorem ipsum dolor sit amet
Consectetur adipiscing elit
Integer ornare lectus ut arcu
Tincidunt porttitor
Feature Title
Lorem ipsum dolor sit amet
Consectetur adipiscing elit
Integer ornare lectus ut arcu
Tincidunt porttitor
Feature Title
Lorem ipsum dolor sit amet
Consectetur adipiscing elit
Integer ornare lectus ut arcu
Tincidunt porttitor
Feature Title
Lorem ipsum dolor sit amet
Consectetur adipiscing elit
Integer ornare lectus ut arcu
Tincidunt porttitor
Feature Title
Lorem ipsum dolor sit amet
Consectetur adipiscing elit
Integer ornare lectus ut arcu
Tincidunt porttitor
Feature Title
Lorem ipsum dolor sit amet
Consectetur adipiscing elit
Integer ornare lectus ut arcu
Tincidunt porttitor
Feature Title
Lorem ipsum dolor sit amet
Consectetur adipiscing elit
Integer ornare lectus ut arcu
Tincidunt porttitor
Feature Title
Lorem ipsum dolor sit amet
Consectetur adipiscing elit
Integer ornare lectus ut arcu
Tincidunt porttitor
Feature Title
Lorem ipsum dolor sit amet
Consectetur adipiscing elit
Integer ornare lectus ut arcu
Tincidunt porttitor
Feature Title
Lorem ipsum dolor sit amet
Consectetur adipiscing elit
Integer ornare lectus ut arcu
Tincidunt porttitor
Feature Title
Lorem ipsum dolor sit amet
Consectetur adipiscing elit
Integer ornare lectus ut arcu
Tincidunt porttitor
Feature Title
Lorem ipsum dolor sit amet
Consectetur adipiscing elit
Integer ornare lectus ut arcu
Tincidunt porttitor
Feature Title
Lorem ipsum dolor sit amet
Consectetur adipiscing elit
Integer ornare lectus ut arcu
Tincidunt porttitor
Feature Title
Lorem ipsum dolor sit amet
Consectetur adipiscing elit
Integer ornare lectus ut arcu
Tincidunt porttitor

As part of the de.KCD/de.NBI network the Center of Digital Health organizes this year’s de.KCD Industry Forum on June 18, 2026 at the Einstein Center Digital Future in Berlin. de.KCD creates a forum

Our colleagues from Northestern University in Boston are organising the 18th INTERNATIONAL CONFERENCE ON SYSTEMS BIOLOGY OF HUMAN DISEASE – SBHD 2026 in Boston from June 08-10. This years Conference

The Berlin Institute of Health at Charité (BIH) coordinates the pan-European research initiative "EU-CiP" (European Cancer Information Portal). Supported by 12 million euros from the EU’s Horizon

The Digital Health Center once again organises the INTERNATIONAL CONFERENCE ON SYSTEMS BIOLOGY OF HUMAN DISEASE – SBHD 2025 in Berlin from June 16-18. Don’t miss this opportunity to participate in a
The Hub for Innovations in Digital Health (HiDiH) brings together two independent sites of excellent research and development in Berlin and in Heidelberg. HIDIH’s major branch in Berlin is the Center for Digital Health at the Charité and the Berlin Institute of Health (BIH).
If we caught your attention, you are interested in our work and would like to get in touch with us, please contact us via franziska.mueller@bih-charite.de



